Transform Annual Product Quality Review & Periodic Review into Continuous Quality Intelligence
AmpleLogic APQR is cloud-based annual product quality review software that automatically aggregates manufacturing, quality, and laboratory data across systems to generate compliant PQR reports and Quality Management Reviews (QMR) — delivering real-time pharmaceutical product quality review insights, process capability analytics, and regulatory inspection readiness.
- Reduce APQR & periodic review preparation time by 70–80%
- Eliminate manual data compilation with paperless quality review
- Ensure GMP compliance across global regulatory bodies
- Enable proactive quality monitoring and digital quality transformation
70-80%
Prep Time Reduction
0
Manual Data Compilation
100%
Audit Trail Coverage
6σ
Statistical Reports
Challenges in Pharma Product Quality Review (PQR) Process
Traditional PQR workflows face critical obstacles that delay reviews, increase compliance risk, and limit data-driven quality improvements
Fragmented Data Collection and Integration
Quality data is spread across LIMS, ERP, QMS, spreadsheets, and paper records, making data aggregation slow, manual, and error-prone, especially in multi-site and legacy environments.
Data Accuracy and Consistency Issues
Manual data transfers, repeated file copies, and static charting increase the risk of inconsistencies, version mismatches, and formatting errors — with manual copy-paste between systems introducing transcription errors that consume review time and drive rework.
Regulatory Compliance Pressure
Evolving regulatory expectations from global authorities demand complete traceability, reproducible analytics, and inspection-ready documentation, which are difficult to maintain with manual PQR processes.
Limited Analytical and Trending Capabilities
Traditional PQR approaches rely on manual charting in tools like Minitab — Exporting to Excel files, creating control charts by hand, and pasting static images into reports — lacking advanced statistical analysis, live control charts, and cross-product trending.
Inefficient Cross-Functional Collaboration
Disconnected workflows across manufacturing, QA, QC, and regulatory teams cause delays, poor visibility into actions, and inconsistent reporting across manufacturing sites throughout the PQR lifecycle.
Weak Audit Trail & Poor Traceability
Traceability to past APQR items is limited and inconsistent. No systematic linkage between current findings and previous open items, making it difficult to demonstrate compliance during regulatory inspections.
70–120 Hours Manual Effort Per Product
Approximately 80% of effort is consumed in data collection, collation, and report compilation — leaving limited time for meaningful analysis, root cause investigation, and continuous improvement.
Cross-Product Analysis Complexity
Lack of linkage between parent and child products makes comparative reviews difficult and time-consuming across product families, increasing the risk of missed systemic quality trends.
Product Quality Review Software for Pharma
Centralize data, automate charts, embed electronic APQR with traceable evidence — delivering comprehensive pharmaceutical product quality review and formatted output dossier assembly
Data Ingestion & Cross-System Connections
Automated pulls from LIMS, ERP, QMS, RIM, and clinical systems through cross-system connections — ensuring a single source of truth with supplier change notification tracking, file uploads, validation, and complete audit trail.
Batch Analysis & Charting
Automated batch analysis software with statistical analyses and control charts using R tools, replacing manual Minitab uploads. Includes process validation software capabilities for CPP/CQA evaluation.
APQR & Periodic Review Workflow
Embedded electronic APQR document with relationship automation that auto-links quality events, CAPAs, deviations, and change controls — plus history of previous APQR open items for full traceability.
Action Tracking & Effectiveness Checks
Create, assign, and track corrective actions with automated effectiveness check processing, 5 Whys root cause analysis, role-based access, and complete audit trail.
Formatted Output & Dossier Assembly
Configurable formatted output templates, automated binder/dossier assembly, PQR report generation, executive summaries, and dashboards for exception-based review.
CPV & Process Validation
Built-in Continued Process Verification monitoring and process validation software with automated statistical analysis, validations, and quality trend analysis.
Multi-Site Quality Governance
Harmonized APQR & QMR templates across all sites with cross-site product comparisons, central quality dashboards, supplier periodic review (SPR) tracking, and global product lifecycle monitoring.
Features of AmpleLogic APQR
AmpleLogic APQR provides configurable APQR program templates that can be defined based on product type (tablet, injectable, API, biologic), manufacturing site, and regulatory market (US, EU, India, Japan). The platform includes built-in template version management, enabling organizations to maintain standardized and compliant review structures across multiple products and global markets. Configurable workflows allow organizations to define review and approval processes involving cross-functional stakeholders such as Manufacturing, QA, QC, and Regulatory teams, ensuring controlled collaboration and full audit traceability.
APQR Template Configuration
APQR Program Templates are configurable based on product type (tablet, injectable, API, biologic), manufacturing site, and regulatory market (US, EU, India, Japan). They include built-in template version management and allow configuration of review and approval workflows for the APQR process.
APQR Template Version Management
Manage the PQR templates based on the Product type and the versions based on the revisions.
APQR Document Version Control
Enable automatic versioning of APQR and PQR documents to maintain traceability and compliance.
Master Data Management
Maintain centralized master data for all quality and process parameters, including limits such as NLT, NMT, Min, Max, Equal To, Descriptive, and Report Value.
Any Time Automated PQR Report Generation
Generate Automated APQR and PQR reports on demand based on selected review periods using standardized, configurable templates.
Automated PQR Execution
Streamline the entire APQR process through automation, reducing manual effort while improving compliance and efficiency.
Six-Pack Report Generation
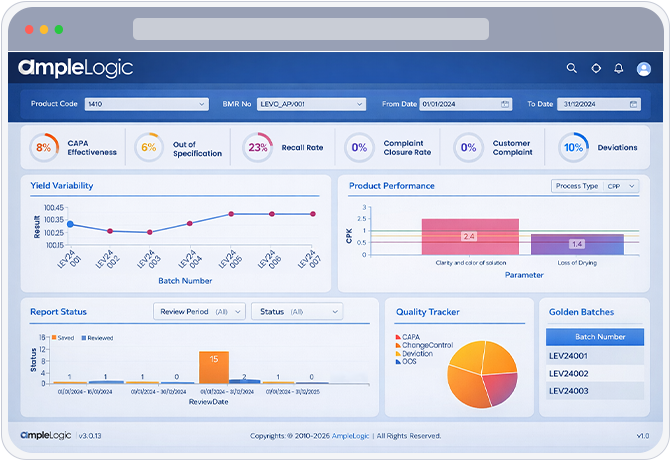
Automatically generate six-pack reports using 3 Sigma and 6 Sigma performance indices for robust process capability evaluation.
Batch-Wise Trend Reporting
Generate batch-wise trend reports to monitor granulation, compression, coating, and packing yields across the review period.
APQR Replaces Statistical Tools
Eliminate dependency on external statistical tools such as Minitab, SAS, or SPSS through built-in analytics, R tools, and reporting.
CPK Assessment with Visual Indicators
Provide color-coded Cpk indicators to quickly assess process capability and identify areas requiring attention.
Nelson Rule Implementation
Apply Nelson's Rules to detect non-random patterns and out-of-control conditions, strengthening CPV reporting.
Critical Control Point (CCP) Monitoring
Define tolerance limits for CCPs and automatically escalate excursions beyond Proven Acceptable Range (PAR) or Normal Operating Range (NOR).
Continued Process Verification (CPV)
Enable continuous monitoring of critical quality and process parameters with automated statistical evaluation to support ongoing compliance.
Golden Batch Facilitation
Support Golden Batch identification by analyzing historical performance and enabling consistent replication of optimal manufacturing conditions.
Process Parameter Comparison Reports
Generate graphical comparisons of process parameters to evaluate the impact of CAPA actions, vendor changes, or process improvements.
Auto Alerts and Warnings
Trigger automated alerts for review delays beyond seven days and warnings for deviations, OOS, and OOT scenarios.
Cross-Functional Collaboration
Facilitate collaboration across Manufacturing, QA, QC, and Regulatory teams through shared workflows and visibility.
Audit Preparedness
Ensure inspection readiness with accurate, compliant APQR, PQR, and Continued Process Verification (CPV) reports supported by traceable data, version history, and audit trails.
Batch Release Check with APQR Results
Enable real-time batch release decisions driven by APQR insights, including pending deviation status and product quality parameter limit checks.
APQR Predictive Insights, Not Just Reports
Leverage built-in statistical tools, batch analysis software, and process validation capabilities to monitor product performance and identify systemic risks before they impact compliance or supply
Process Capability & Validation Analysis (Cp, Cpk)
Calculate Cp, Cpk, Pp, Ppk, and standard deviation automatically for QA decisions. Complete process validation software capabilities for process capability assessment.
Control Charts & Quality Trend Analysis
Visual evidence for process stability with I-charts showing control limits, specification limits, and statistical trend lines. Comprehensive quality trend analysis across all parameters.
Out-of-Specification Trend Monitoring
Automated OOS/OOT detection and trending across all parameters, flagging excursions before they impact drug product quality review outcomes.
Deviation & Complaint Correlation
Cross-reference deviations, complaints, and CAPAs with relationship automation to identify systemic risks and root cause patterns across products and manufacturing sites.
Stability & Yield Performance Analytics
Monitor long-term stability trends and pharmaceutical manufacturing software analytics to detect yield degradation and batch analysis anomalies early.
Six-Pack Reports & R-Based Analysis
Generate comprehensive six-pack reports with I-charts, normal probability plots, batch variation, and embedded R analysis — replacing manual Minitab charting entirely.
AI-Powered PQR Data Extraction, Root Cause Analysis & Quality Insights
Faster, more accurate PQR report generation with live QA insights, OCR extraction, automated language translation, quality incident triage, and AI chatbot support
OCR Data Extraction
Automates extraction from scans, handwriting, and PDFs. Minimizes manual errors and enforces uniformity across all data sources for paperless quality review.
AI ChatBot for Query Resolution
Instantly access historical quality data to identify process deviations, perform root cause analysis, and get immediate answers to report-related questions.
AI-Based Summary & 5 Whys Analysis
Get section-wise automated summary and conclusions in the final report, plus AI-assisted 5 Whys root cause analysis — reducing manual narrative writing by 90%.
Faster Report & Dossier Generation
Reduce report time from hours to minutes with automated data aggregation, chart generation, formatted output templates, and binder/dossier assembly.
Quality Incident Triage & Alerts
Intelligent quality incident triage to determine escalation need, with real-time QA alerts for immediate corrective action and oversight across all parameters.
Automated Language Translation
Multilingual support with automated language translation for complaint processing, data translation, and global team collaboration across manufacturing sites.
Enterprise Data Integration & Cross-System Connections
AmpleLogic APQR integrates seamlessly with enterprise systems to create a single source of truth for product quality. The platform automatically consolidates In-Process Parameters and Critical Process Parameters (CPPs) from Batch Manufacturing Records, process control and analytical parameters, stability data from QC laboratories, and quality events such as deviations, CAPA, and change controls. This unified view enables comprehensive, data-driven Annual Product Quality Reviews. Detailed integration parameters are listed below by process area.
RIMS
Cross-system Quality-RIM connection for regulatory data sharing and compliance reviews
eQMS
Deviations, CAPAs, complaints, change controls, and continuous improvement records
LIMS
Lab incidents, OOS/OOT data, and analytical results
MES / eBMR
Production QMS data, batch records, and pharmaceutical manufacturing data
DMS
New and updated documents linked to reviews
Supplier
Supplier periodic review (SPR), supplier change notifications, and vendor quality data
eLOG
Logbook data from manufacturing operations
CAPS
Calibration, instrument records, and audit program planning data
ERP
External system data and supply chain information
Stability
Stability study data for trending and shelf-life analysis
Result: A unified quality dataset for faster and more reliable product reviews
APQR Regulatory Compliance: FDA, EU GMP & ICH-Ready Quality Reviews
AmpleLogic APQR supports regulatory expectations across major health authorities worldwide
Supported Health Authorities
FDA
U.S. Food & Drug Administration
EMA
European Medicines Agency
MHRA
UK Medicines & Healthcare products
WHO
World Health Organization
CDSCO
Central Drugs Standard Control
Platform Compliance Features
21 CFR Part 11 & GMP Compliance
Electronic Signatures with Change Justification
Complete Audit Trails & Reasons for Change
Validation-Ready Architecture (IQ/OQ/PQ)
Role-Based Access Control & Quality Teams
Regulatory alignment: FDA 21 CFR 211, EU GMP Annex 15, ICH Q7, ICH Q10, WHO TRS, and regional health authority requirements. Full GMP compliance software with reasons for change tracking.
APQR Software ROI: Measurable Results for Enterprise Quality Assurance
Organizations using AmpleLogic APQR achieve transformational outcomes across quality operations
70% Reduction
In manual APQR preparation effort
Faster Inspections
Regulatory inspection readiness improved significantly
Cross-Functional Collaboration
Improved collaboration between QA, QC, manufacturing, and regulatory teams
Early Trend Detection
Proactive identification of product quality trends before they become compliance issues
APQR Software Customer Success Stories
Multi-site APQR and CPV/OPV implementations, harmonizing global templates while supporting site-specific data models, workflows, report formats, and statistical requirements.
Digital PQR and CPV implemented across sites with automated report generation, covering APQR, CPV, trending, and stability summaries, with dynamic templates aligned to customer SOPs and regulatory expectations.
Why AmpleLogic's APQR?
AmpleLogic's AI-powered APQR solution is purpose-built for pharmaceutical companies, API manufacturers, CDMOs, Biotech, Biosciences, and CROs. This advanced SaaS-based platform is designed in alignment with US FDA and GMP requirements and complies with ICH Q7 (Section 2.5), which mandates periodic quality reviews of APIs, referred to as Product Quality Reviews (PQRs) under EU GMP guidelines.
The solution streamlines PQR generation through intelligent analytics, end-to-end process tracking, and dynamic data visualization to support regulatory compliance, operational efficiency, and quality oversight. Transform annual product quality reviews into an Anytime PQR capability, where automated data aggregation, analytics, and dashboards provide continuous insights into product quality and compliance readiness.
It enables continuous monitoring of Critical Control Points (CCPs) with automated alerts for deviations beyond Proven Acceptable Range (PAR) and outside Normal Operating Range (NOR). Quality teams can easily generate detailed three-sigma and six-sigma reports with real-time notifications, ensuring consistent compliance, faster decision-making, and sustained product quality.
Cloud-Based SaaS Quality Platform
Cloud-based APQR software and PQR software for pharma with SaaS quality management, zero infrastructure overhead, automatic updates, and enterprise-grade security.
100% Customizable & Configurable
Tailor workflows, formatted output templates, and interfaces to current operations with full configurability — no rigid vendor-dictated processes.
GMP Compliant with Major Regulators
Prebuilt controls and GMP compliance software for USFDA, MHRA, WHO, ICH Q7, and ICH Q10 validations with regulatory inspection readiness.
10x Faster Deployments
Accelerated rollout and time to value versus competitors with pre-validated templates and digital quality transformation methodology.
Continuous Product Upgrades
Regular updates with validation-ready change controls, continuous improvement features, and ongoing support.
Industries We Serve
Trusted across pharmaceutical, biotech, CDMO, API manufacturing, and life sciences organizations
Pharmaceutical Manufacturing
Monitor batch yield, deviations, stability trends, and complaints using annual product quality review software for comprehensive pharmaceutical product quality review and regulatory dossier generation.
Biotech & Biologics
Aggregate complex process and stability data for biologics and advanced therapies with specialized batch analysis and process validation analytics.
Contract Manufacturing (CDMO)
Generate APQR and periodic review reports for multiple clients with standardized GMP compliance workflows and client-specific formatted output templates.
API Manufacturers
Active pharmaceutical ingredient quality reviews with process capability tracking, impurity trending, and ICH Q7 compliance for drug product quality review.
Biosciences & CROs
Research-grade product quality reviews with multi-client analytical data management, statistical analysis, and Quality Management Review (QMR) capabilities.
Medical Devices
Device quality reviews with performance trending, complaint analysis, supplier periodic review (SPR), and regulatory compliance tracking.
"Providing auto-generated trend charts with statistical analysis. This software is a time saving activity for each employee. Truly a game changer for PQR preparation."
Maya K.
Quality Analyst
"Auto generated APQR format with built-in trend charts, Cp/Cpk calculations, and statistical outputs. Time saving and highly compliant for pharma quality reviews."
Sarita A.
QA Manager
"The integration with LIMS and stability data reduces manual data entry significantly. Automated report generation ensures consistency across all product reviews."
Verified G2 User
Pharma Quality Lead
See How Global Pharma Teams Automate Product Quality Reviews
Transform manual, error-prone processes with AmpleLogic APQR software — the leading PQR software for pharma that delivers automated, compliant quality intelligence, 70–80% reduction in preparation effort, and digital quality transformation.
Stay Ahead in Life Sciences
Get the latest product updates, compliance news, and industry insights delivered to your inbox.